-40%
CE ECG600G EKG Machine ECG Electrocardiograph 6 Channel Touch 12 Lead+PrinteR+SW
$ 242.35
- Description
- Size Guide
Description
CE ECG600G EKG Machine ECG Electrocardiograph 6 Channel Touch 12 Lead+PrinteR+SWContec Medical Systems---ECG600G 6 Channel ECG Machine
Introduction
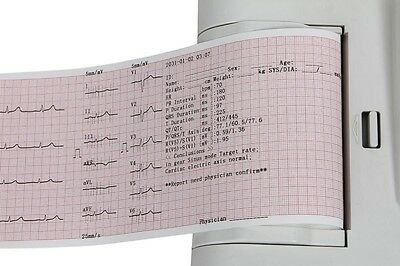
ECG600G is an Electrocardiograph which can collect 12-lead ECG signal and print waveform by the thermal printing system. It features in, recording and displaying ECG waveform in AUTO/Manual mode, measuring and diagnosing ECG waveform parameters automatically, prompting for “Lead off” and “Lack of paper”, multi-language interface, case database management.
Function
1)Adopt high-resolution thermal printing system(8 dot/mm), no need for any adjustments. Recording frequency: up to 150Hz.
2)Real-time and continuously record the clear and accurate ECG waveform and annotation character(including lead mark, sensitivity, paper speed and filter state, etc.).
3)Full-digital filter design, anti-baseline drift, AC and EMG interference.
4)Adjust the baseline automatically, optimize the print setting and optional rhythm leads.
5)In AUTO mode, finish recording by pressing the button once, which improves work efficiency.
6)Adopt digital signal processing technology and get high-quality ECG waveform via power frequency filter, baseline filter and EMG filter of ECG signal.
7)With the functions of auto-measurement, auto-analysis and auto-diagnosis for routine ECG parameters, which reduces the doctor’s burden and improves work efficiency.
8)Sync collection for 12-lead ECG, multiply printing formats, including 2×6+1(rhythm lead), 2×6, 3×4, 3×4+1(rhythm lead), 4×3, 4×3+1(rhythm lead), 6×2, 6×2+1(rhythm lead), rhythm 2, rhythm 3, rhythm 4, rhythm 5, rhythm 6, rhythm 7, channel record, etc.
9)Display of 3/6/12-lead ECG, "Lead off" and "Lack of paper" on one screen, and with ECG DEMO mode. Auto-check and auto-analysis for ECG parameters, ECG waveform can be recorded manually or automatically. And patient information can be stored by case database.
10)Time print can be set to every 1/2/3/5/10/20/30/60 minutes.
11)Print setting, including “Print and save”(print waveform and save case), “Print no save”(only print waveform but not save case), “Save no print”(only save case but not print waveform).
12)AC/DC, built-in rechargeable lithium battery and auto-protection circuit, and automatic power off when there is no operation in appointed time.
13)Store more than 1000 cases, convenient for case review and statistic.
14)Multi-language(Chinese, English, French, Spanish, Turkish, Polish, Italian, German, Portuguese, Kazakstan, Russian and Ukrainian) interface and report.
Performance
Input mode: floating and defibrillation protection
Frequency response: 0.05Hz ~ 150Hz(-3dB~+0.4dB)
CMRR: >60dB, >100dB(add filter)
Time constant:≥ 3.2s
Patient leak current: <10µA
Calibration voltage: 1mV
Sensitivity: 2.5, 5, 10, 20, 40 mm/mV ±5%, standard sensitivity: 10mm/mV±2%
Noise level: <15µVp-p
Input circuit current: ≤50nA
Input impedance: ≥50MΩ
Sampling accuracy: 12-bit
Safety classification: class I, type CF and defibrillation-proof applied part
Recording mode: thermal printing system
Auto record: set record according to auto record format and mode, automatically switching leads, measuring and analyzing.
Rhythm record: set record according to rhythm record format and mode, automatically measuring and analyzing.
Manual record: set record according to record format, manually switching leads.
Measurement parameters: HR, P-R interval, P Duration, QRS Duration, T Duration, Q-T interval, Q-Tc, P Axis, QRS Axis, T Axis, R(V5), S(V1), R(V5)+S(V1)
Paper size: 80mm(W)×20m(L)
Sampling frequency: 1000Hz
EMG interference filter: 25Hz/35Hz (-3dB)
Power frequency filter: AC 50Hz/60Hz(-20dB)
Paper speed:
Auto: 5, 6.25, 10, 12.5, 25, 50, 100mm/s, ±5%
Rhythm: 5, 6.25, 10, 12.5, 25, 50, 100mm/s, ±5%
Manual: 5, 6.25, 10, 12.5, 25, 50, 100mm/s, ±5%
Accessories
Standard:
A lead cable
A limb electrode
A chest electrode
A thermal recording paper
A power cord
An earth wire
A user manual
ECG-Sync software+USB cable
Optional:
ECG electrode
Bag
Conductive gel
Physical characteristic
Dimension: 315mm(L) × 215mm(W) × 92mm(H)
Weigh: 1.7Kg
* We accept payment from PAYPAL,
* All payments are expected within 7 days after the last winning auction is closed.
* Clearance: we will ship your item(s) to your confirmed address.Please leave me your clear address,telephone,postal code.
* It is the buyers' responsibility to consider duties, taxes and other extra charges by the government in your respective country.
Buy safe Products
The following FDA Disclaimer is required for all eBay listing in Healthcare category and is included for REFERENCE:
The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies.If the item is subject to FDA regulation, We will verify your status as an authorized purchaser of this item before shipping of the item.
If you have questions about legal obligations regarding sales of medical devices, you should consult with the FDA's Center for Devices and Radiological Health.
The Fingertip Pulse Oximeter is registered on the Australian Register of Therapeutic Goods (ARTG) with the code 197923,
and certified by FDA of United States and CE,TUV of Europe.The Fingertip Pulse Oximeter that is FDA 510K Approved
Contec Medical Systems focusing on research, manufacture and distribution of medical instruments,
was founded in 1992 as a high-tech company.
At present there are more than 1200 employees in our company.
Our product line covers a wide range of 13 categories.
Most of the domestic hospitals are our
customers.
Contec hopes to cooperate
with international companies to supply more
innovative design and
advanced technology products.
We sincerely welcome you to become
one of our global partners.
We are looking forward to establishing a successful business
relationship with you.
Contec Medical Systems---Skype contec.tessy, contecmxf#gmail.com
Wechat/Whatsapp/Mobile:86-18716007715
Only English user guide, if you need any other language, please contact us!
Only English user guide, if you need any other language, please contact us!